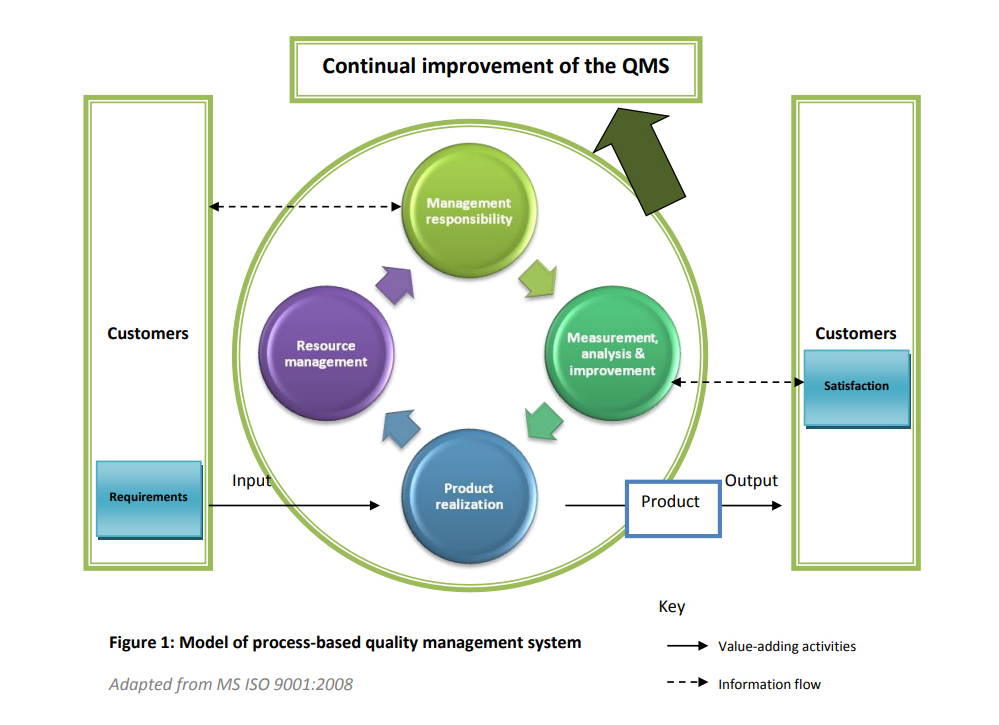
Quality Management System
Training Program Outline
- ISO 9001 Quality Management System Requirements Training
- ISO 9001 Quality Management System Documentation Requirements Training
- ISO 9001 Quality Management System Internal Quality Auditor Training
Overview
Any business that wants to stay in business has to keep up by offering quality products and services as well as providing assurance of this commitment. Organizations can demonstrate this and secure themselves a future through an ISO 9001 Quality Management System (QMS) certification.
This standard is recognized worldwide in more than 140 countries and belongs to the ISO 9000 series of standards. The ISO 9000 series is amongst the International Organisation for Standardisation’s (ISO) most widely known and used standards.
ISO 9001 has been designed to be a ‘generic’ management system standard, applicable for organizations of any size and type. Thus, it is widely accepted and adopted by manufacturing and service organisations, non-profit organizations, and even governmental departments the world over.
ISO 9001 provides an effective framework for any quality conscious organisation that would like to consistently provide products and services that meet their customers’ requirements; and continually improve on their products, processes and system to enhance satisfaction.
ISO 9001:2008 lays down what requirements your quality system must meet, but does not dictate how they should be met in any particular organization. This leaves great scope and flexibility for implementation in different business sectors and business cultures, as well as in different national cultures.
Objectives :
- To establish a quality management system in meeting customer requirement
Benefits of Certification :
- A systematic approach to control all its processes and well defined procedures and supporting documentation
- Greater transparency and consistency in its output
- An established plan-do-check-act (PDCA) approach that drives improvements
- Greater quality awareness amongst employees and reduced defects, scraps, rework, failures and service recovery
- The means to assure a sound quality management system by having a third party certification that is recognised worldwide
- Correspondingly, a reduced need for multiple second party audits
- And ultimately, a reduction in customer complaints, retention of customers and an increase in its ability to penetrate new markets and increase its market share
- Prove your commitment to quality and customer satisfaction
- Ensures your products and services effectively meet customer and applicable statutory and regulatory requirement
Environmental Management System
Training Program Outline
- ISO 14001 EMS Requirements
- ISO 14001 EMS Aspect and Impact Analysis
- ISO 14001 Chemical Management & Waste Minimization
- ISO 14001 EMS Emergency Preparedness
- ISO 14001 EMS Environmental Laws
- ISO 14001:2004 Internal Auditing
- ROHS Directive
Overview
An EMS meeting the requirements of ISO 14001:2004 is a management tool enabling an organization of any size or type to:
• identify and control the environmental impact of its activities, products or services, and to
• improve its environmental performance continually, and to
• implement a systematic approach to setting environmental objectives and targets, to achieving these and to demonstrating that they have been achieved.
ISO 14001:2004 does not specify levels of environmental performance. If it specified levels of environmental performance, they would have to be specific to each business activity and this would require a specific EMS standard for each business. That is not the intention.
ISO has many other standards dealing with specific environmental issues. The intention of ISO 14001:2004 is to provide a framework for a holistic, strategic approach to the organization’s environmental policy, plans and actions.
ISO 14001:2004 gives the generic requirements for an environmental management system. The underlying philosophy is that whatever the organization’s activity, the requirements of an effective EMS are the same.
This has the effect of establishing a common reference for communicating about environmental management issues between organizations and their customers, regulators, the public and other stakeholders.
Because ISO 14001:2004 does not lay down levels of environmental performance, the standard can to be implemented by a wide variety of organizations, whatever their current level of environmental maturity. However, a commitment to compliance with applicable environmental legislation and regulations is required, along with a commitment to continual improvement – for which the EMS provides the framework.
Benefits of Certification
- Improved corporate image among regulators, customers and the public
- Improved perception and understanding of key environmental issues by all within the organisation
- A framework for continuous improvement of the organisation’s environmental performance and an improved ability to comply with regulatory requirements
- Measures taken to generate less waste will lead to reduced product, material handling and waste disposal costs that can be ploughed back into the business (improving cost control through conserving input material and energy).
- Increased efficiency and better use of energy and raw materials will invariably lead to savings and a more profitable business
- A strong environmental image can help to attract environmentally conscious customers and in this, expand market share
- Increased awareness and environmentally conscious actions will lead to betterment of the environment for the community and the nation at large
Occupational Health & Safety System
Training Program Outline
- Occupational Health and Safety (OHSAS 18001) New Requirements
- OHSAS 18001 Related Laws & Regulations
- Identification, Assessment and Risk Control of Hazardous Substances
- Chemical Management
Overview
Organizations can better address occupational health and safety (OH&S) concerns in a more orderly and consistent manner with the implementation of an OH&S Management System (OHSMS). The OHSAS 18001 OHSMS provides a framework for organizations to examine issues such as the allocation of resources, assignment of responsibilities and on-going evaluation of practices, procedures and processes systematically, and to achieve continuous improvement in OH&S performance through effective management of their risk impacts.
The current specification is based on the British Standard Institution’s (BSI) OHSAS 18001, an international Occupational Health & Safety Management System specification that has been widely adopted by many countries in the world for their OH&S Management System certification. The main intent of this management system standard is to control risks and prevent harm to those at work.
With the OHSAS 18001 Occupational Health and Safety (OH&S) Management System, an organisation is able to manage its OH&S risks with the same degree of expertise and control as its other business activities. The system is practical and takes into account the organisational structure, planning activities, responsibilities, practices, processes and resources for developing, implementing, achieving, reviewing and maintaining the organisation’s OH&S policy.
Benefits of Certification:
- Improve efficiency and consequently reduce accident and production time loss
- Increase control and reduction of hazards through the setting of objectives, targets and evolved responsibility>>reduction of safety related costs
- Demonstrate your commitment to the protection of staff, property and plant
- Demonstrate legal compliance
- Increase your reputation for safety and occupational health
- Reduce insurance premiums
- Is an integral part of a sustainability strategy
- Encourage more effective internal and external communication
- Improve your safety culture
Medical Device Directives
Training Program Outline
- ISO 13485 Medical Devices Directive Requirements
- ISO 13485 Internal Auditor
- Good Manufacturing Practice for Pharmaceutical Industry
Overview
ISO 13485 specifies requirements for a quality management system that can be used by an organization for the design, production, installation and servicing of medical devices. It is based on ISO 9001:2000 quality management standard but it does not include the requirements related to continual improvement and customer satisfaction. At the same time, it requires additional documented procedures that consistently meet customer and regulatory requirements applicable to medical devices and related services.
Benefits of Certification:
- Assures authorities that a manufacturer of medical services has developed and implemented a quality management system based on a widely accepted International Standard.
- Demonstrates a manufacturer’s ability to supply medical devices and related services to customers that comply with regulatory requirements.
- Facilitates the export of medical services to markets where regulatory requirements apply.
- A reduction in customer complaints and retention of customers
Food Safety Management System
| ISO 22000 | HACCP | GMP |
| HALAL | Food Handler Training | Others |
Training Program Outline
- ISO 22000 Management System Requirements
- ISO 22000 Internal Auditor
Overview
The ISO 22000 is an auditable standard which integrates the seven principles of the Hazard Analysis and Critical Control Points (HACCP) system and application steps developed by the CODEX Alimentarius Commission. It is applicable to all organizations, regardless of size, which are involved in any aspect of the food chain and want to implement systems that consistently provide safe products. The means of meeting any requirements of ISO 22000:2005 can be accomplished through the use of internal and/or external resources.
The ISO 22000 international standard specifies the requirements for a Food Safety Management System (FSMS) that involves interactive communication, system management, prerequisite programmes and HACCP principles. It is relevant to all parties in the food chain as food safety hazards can occur at any stage in the food chain and a combined effort of all parties is required.
ISO 22000 requires that all hazards that may be reasonably expected to occur in the food chain, including hazards that may be associated with the type of process and facilities used, be identified and assessed. Hazard analysis helps in establishing the hazards that need to be controlled and those that need not. Subsequently, the organization is able to determine an effective combination of control measures and come up with the prerequisite programmes and the HACCP plan.
Benefits of Certification:
- Greater impact on customers
- Increased transparency
- Streamlined production
- Minimization of significant food risk
- Effective control of internal processes and minimizing risk of failure
- Increased staff motivation by focusing on a job well done
- Signal sent about a proactive approach to food safety
- Focus kept on your essential challenges
Training Program Outline
- HACCP Requirements
- HACCP Internal Auditor
Overview
Hazard Analysis and Critical Control Point (HACCP) certification is synonymous with food safety. HACCP is a food safety risk management system that addresses biological, chemical and physical hazards through anticipation and prevention rather than by a finished product inspection. The Good Manufacturing Practice (GMP), or Good Hygiene Practice (GHP), or Good Agriculture Practice (GAP) certifications are required prerequisites for an effective HACCP system.
HACCP is an internationally recognized system that assures food safety at all stages of the food chain and provides a more structured approach to the control of food hazards compared to the traditional inspection and quality control procedures. Its emphasis is on prevention and hence, the control of hazards is built into the design, manufacturing, packaging and distribution as well.
Benefits of Certification:
- A sound risk-assessment framework that allows both industry and government to allocate their resources efficiently in establishing and auditing safe food production practices
- An internationally-recognised system for assuring food safety
- Improved compliance with hygiene regulation
- Customer recognition and approval, as well as the potential to penetrate new markets
Training Program Outline
- GMP Requirements
Overview
Good manufacturing practice (GMP) is a system for ensuring that food is not adulterated, is safe for human consumption and consistently produced and controlled in accordance to established quality and food safety standards. Moreover, GMP is designed to minimize risks in any food production that cannot be eliminated through testing of the final product.
GMP regulations address issues including recordkeeping, personnel qualifications, sanitation, cleanliness, equipment verification, process validation, and complaint handling. Most GMP requirements are very general and open-ended, allowing each manufacturer to decide individually how to best implement the necessary controls. This provides much flexibility, but also requires that the manufacturer interpret the requirements in a manner which makes sense for each individual business.
GMP is comprehensive and covers all aspects of production: from materials, to premises, equipments, handling practices, processing, packaging, storage and delivery, even to the training and personal hygiene of staff.
The GMP to be implemented depends on which segment of the food chain that the organization operates. Some examples of equivalent terms used are: Good Agricultural Practice (GAP), Good Veterinarian Practice (GVP), Good Hygienic Practice (GHP), Good Production Practice (GPP), Good Distribution Practice (GDP) and Good Trading Practice (GTP).
GMP is an absolute essential as it provides the foundation for the development and implementation of successful Food Safety Management Systems (FSMS) as well as an assurance of food safety. The upshot is, GMP is a prerequisite for all who are committed to food safety and FSMS standards such as Hazard Analysis and Critical Control Points (HACCP) and ISO22000.
Benefits of Certification:
- builds consumers’ confidence that food is produced in accordance to the best manufacturing practices
- easier to obtain HACCP certification
- enhance image of company in the production of safe food and increase competitive edge in the international market.
- Facilitate the process of meeting the requirements of Malaysian Food Act 1983 and Malaysia Food Regulations 1985.
Training Program Outline
- HALAL Requirements
Overview
Halal is an Arabic word means lawful or permitted. This term can be used in relation to food products, meat products, cosmetics, personal care products, pharmaceuticals, food ingredients, and food contact materials. Although halal food or services are readily available and easy to obtain, there is a need for the authority to monitor and enforce standards to ensure halalness particularly when the products or services are made, sold, prepared or provided by non-Muslims. Basically, All foods are considered halal except:
- Swine/Pork and its by-products
- Animals improperly slaughtered or dead before slaughtering
- Alcoholic drinks and intoxicants
- Carnivorous animals, birds of prey and certain other animals
- Foods contaminated with any of the above products
Benefits of Certification:
- builds consumers’ confidence that the products are suitable for usage or consumption of Muslim
Training Program Outline
- The characteristics of food poisoning
- The effects of food poisoning on consumers & food business
- Types, sources, vehicles & routes of contamination
- Personal Hygiene & Health
- Controlling food contamination through storage, preparation, cooking & serving
- Methods of cleaning & waste disposal control
- Introductory to Food Safety Systems (GMP, HACCP & ISO22000 FSMS)
- Legal Compliances (Food Act 1983 & Regulation 1985)
Overview
According to Food Hygiene Regulation being enforced since 2009, all food handlers must possess a fundamental knowledge in maintaining good practice in handling, processing and preparation of safe food. This course has been developed in line with Ministry of Health (MOH) recommendation
Non-compliance to the above can results in a fine of not more than RM10,000
OR
not more than 2 years imprisonment
Benefits of FHT:
- To prevent a possible food-borne illness from occurring
- Avoid food poisoning in food premise
- Increase confidence level while facing inspection from local enforcement
| EUROPGAP | Food Labeling | Nutritional Facts |
| Allergen Control | BRC Global Standard | International Food Standard |
| GLP | Basic Food Hygiene & Safety | |
Automotive / Technical Specification
Training Program Outline
- ISO/TS 16949 Requirements
- Core Tools Linkages and Application
- Automotive Process Approach (Customer Oriented Process, Supporting Process,
- Management Process)
- Advanced Product Quality Planning (APQP)
- Failure Mode Effects Analysis (FMEA)
- Statistical Process Control (SPC)
- Measurement System Analysis (MSA)
- Production Part Approval Process (PPAP)
- ISO/TS 16949 Internal Auditing Training
Overview
ISO/TS 16949 is an ISO Technical Specification that specifies the quality system requirements for the design/development, production, and where relevant, installation and servicing of automotive-related products. The standard aims to align the existing automotive quality system requirements within the global automotive industry. It is based on the eight quality management principles : customer focus, leadership, involvement of people, process approach, system approach to management, continual improvement, fact based decision-making, mutually beneficial supplier relationship.
Benefits:
- fulfil one of the conditions of supply to some of the major automotive makers
- gain customer recognition and approval and open doors to the worldwide markets
- have a systematic approach to control all its processes and proven methods that will help to improve product quality levels
- greater transparency and consistency in its output
- and ultimately, a reduction in customer complaints and retention of customers
Quality Improvement Tools / Techniques / Quality Management
Training Program Outline
- Poka Yoke
- 7QC Tools of Effective Problem Solving Techniques
- Advanced 7 QC Tools for Quality Improvement
- Quality Function Deployment
- Quality Control Circle (QCC)
- Cost of Quality
- Design of Experiments
- Eight Disciplines (8D) in Problem Solving
- 5s Housekeeping
- Zero PPM Defect Application
- Quality Management Representative Course
Laboratory System
Training Program Outline
- ISO/IEC 17025 Requirements
- ISO/IEC 17025 Internal Auditor
- Measurement Uncertainty
Overview
ISO/IEC 17025:2005 specifies the general requirements for the competence to carry out tests and/or calibrations, including sampling. It covers testing and calibration performed using standard methods, non-standard methods, and laboratory-developed methods. ISO/IEC 17025:2005 is for use by laboratories in developing their management system for quality, administrative and technical operations.
Benefits
- Having access to more contracts for testing and/or calibration. Some public and private organizations only give contracts to accredited laboratories. Accreditation will also help to get more contracts from organizations that don’t mandate accreditation, but do give preference to accredited laboratories in competitive situations.
- Improved national and global reputation and image of the laboratory.
- Continually improving data quality and laboratory effectiveness.
- Having a basis for most other quality systems related to laboratories, such as Good Manufacturing Practices and Good Laboratory Practices.
Business Improvement Tools & Techniques
Training Program Outline
- Six Sigma Executive Overview
- Champion Training
- Six Sigma Green Belt Course
- Six Sigma Black Belt Course
- Total Quality Management
- Lean Manufacturing / Lean Office
Team Building / Soft Skills Training
Training Program Outline
- Train The Trainer
- Good Tiime Management
- Team Building
- Communication Skills
- Other customised trainings